On 13th October, the U.S. e-cigarette market suddenly came big news.
The world’s first electronic cigarette brand to pass the U.S. PMTA has been born, that is, British American Tobacco’s Reynolds Tobacco (RJ Reynolds) Vuse.
FDA: First-time legal sale of e-cigarettes allowed, suitable for protecting public health
The U.S. Food and Drug Administration today announced it has authorized the sale of three new tobacco products, marking the first set of electronic nicotine delivery system products to receive FDA authorization through the Premarket Tobacco Product Application (PMTA) route.
The FDA issued a marketing authorization order to RJ Reynolds (RJR) Vapor Company for its Vuse Solo enclosed e-cigarette device and accompanying tobacco-flavored e-cigarette cartridges, including the Vuse Solo Power Unit, Vuse Replacement Cartridge Original 4.8% G1, and Vuse Replacement Cartridge Original 4.8% G2.
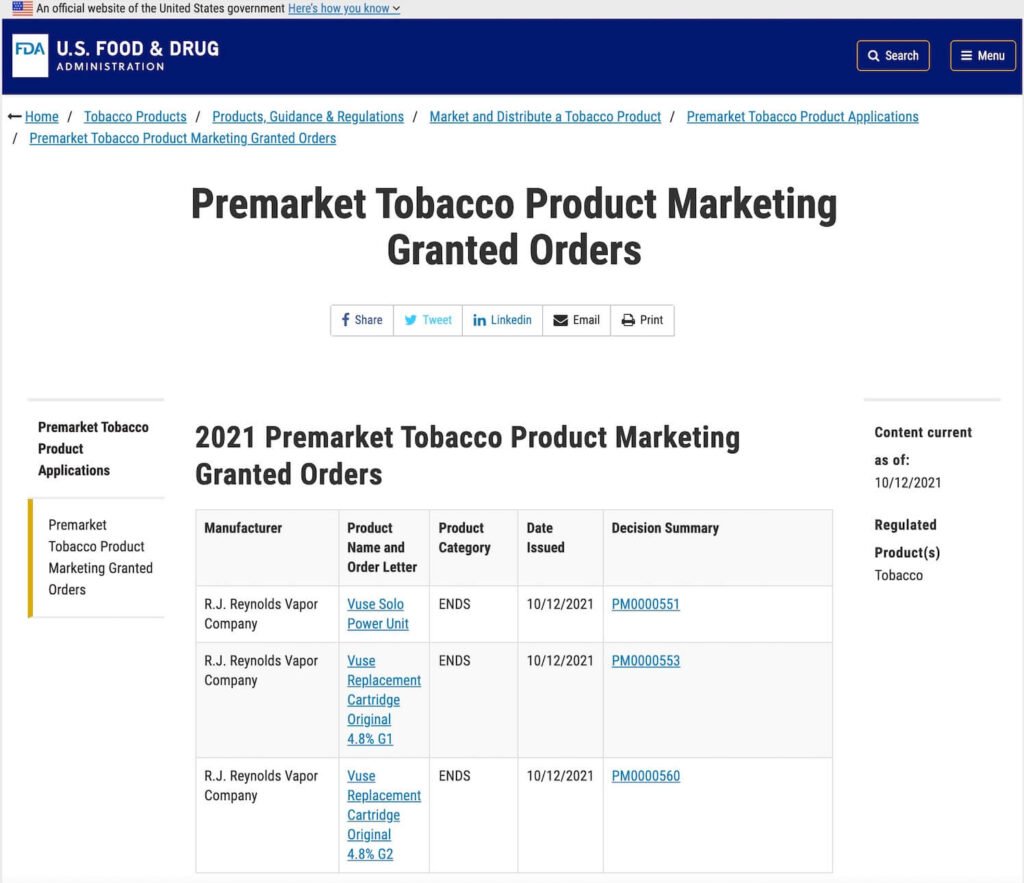
RJR Vapor submitted data to the FDA demonstrating that the marketing of these products is appropriate to protect public health, and the FDA authorized permission for these products to be legally sold in the United States.
“Today’s authorization is an important step in ensuring that all new tobacco products pass the FDA’s robust, science-based premarket evaluation. Data from manufacturers show that their tobacco-flavored products can benefit addicted adult smokers who turn to these products by reducing exposure to harmful chemicals, either completely or significantly, by reducing cigarette consumption.” said Mitch Zeller, director of the FDA’s Center for Tobacco. “We must remain vigilant about this authorization, and we will monitor the marketing of the products, including whether companies are not complying with any regulatory requirements or whether there is reliable evidence of significant use by individuals, including youth, who have not previously used tobacco products, and we will take action as appropriate.”
Under the PMTA pathway, e-cigarette manufacturers must demonstrate to the agency that, among other things, the marketing of new tobacco products is appropriate to protect public health. These products were found to meet this criterion because, among several key considerations, the agency determined that study participants who used only licensed products were exposed to fewer harmful and potentially harmful constituents (HPHC) than users of combustion cigarettes.
The toxicological assessment also found that aerosols of licensed products were significantly less toxic than burning cigarettes, based on comparisons of available data and the results of nonclinical studies. In addition, FDA considered the risks and benefits to the entire population, including users and nonusers of tobacco products. Importantly, this included a review of available data on the likelihood of use of the product by young people. For these products, FDA determined that the potential benefits to smokers who completely or significantly reduce cigarette use would outweigh the risks to youth if the applicant followed postmarketing requirements designed to reduce youth exposure and exposure to the product.
Today, the FDA also issued 10 marketing denial orders (MDOs) for flavored products submitted by RJR under the Vuse Solo brand. The FDA does not publicly disclose specific flavored products due to potential confidential business information issues. These products subject to MDOs for premarketing applications may not be introduced or delivered for introduction into interstate commerce. If any of them are already on the market, they must be removed from the market or executed at risk. Retailers should contact RJR with any questions about the products they have in stock.
The agency is still evaluating the company’s application for the Vuse Solo brand of menthol-flavored products.
The FDA is aware that the 2021 National Youth Tobacco Survey (NYTS) found that approximately 10% of high school students currently selected Vuse as their commonly used brand. The agency takes these data very seriously and considers the risks to youth in its review of these products.
The evidence also suggests that young people are less likely to start using tobacco-flavored products and then move to higher-risk products, such as burning cigarettes, than users of non-tobacco-flavored e-cigarette products.
The data also indicate that most youth and young adults who use e-cigarettes as the beginner using flavors such as fruit, candy, or menthol rather than tobacco flavors.
In addition, today’s mandate imposes strict marketing restrictions on the company, including digital advertising restrictions as well as radio and television advertising restrictions, to significantly reduce the likelihood that youth will be exposed to tobacco advertising for these products.
RJR Vapor Company is also required to report regularly to the FDA about products in the marketplace, including but not limited to ongoing and completed consumer research, advertising, marketing plans, sales data, information about current and new users, manufacturing changes, and adverse experiences.
If FDA determines that continued marketing of a product is no longer appropriate to protect public health, for example, if there is a significant increase in the youth epidemic.
While today’s action allows tobacco products to be sold in the United States, it does not mean that these products are safe or approved by the FDA. All tobacco products are harmful and addictive, and people who do not use tobacco products should not start.
The FDA said the agency has acted on more than 98% of the applications submitted by the deadline. This includes issuing MDOs for more than 1 million flavored e-cigarette products that lack sufficient evidence that adult smokers who use the flavored product will overcome the public health problems associated with the product’s well-documented and considerable appeal to young people.
Recently, the FDA released a sample MDO decision summary. This sample does not reflect the decision basis for each MDO action taken by the FDA.
The agency will continue to issue decisions on applications as appropriate and is committed to transforming the current marketplace into one in which all available e-cigarette products demonstrate that the marketing of the product is appropriate to protect the public health.”
Vuse and IQOS Go Forward Together
With Vuse’s three products passing the FDA’s PMTA review, it means that the e-atomization category of e-cigarettes has a new identity, which also makes Vuse and heating non-burnt e category IQOS side by side, can be said to be the two flowers in the field of atomization and heating.
It is worth noting that the FDA’s first authorization has a very deep significance.
First, Only the tobacco flavor approved, means that all flavored e-cigarettes are unlikely to pass, tobacco-approved is to give adult smokers a choice, refuse to flavored e-cigarettes, is to deny teenagers interested in e-cigarettes, this is a basic principle, all flavored e-cigarette is impossible to pass.
Second, the menthol flavor pending. This also makes people worry, if there are no fruit-flavored e-cigarettes, the mint flavor may become the second-best choice for smokers, if the mint is also banned, then smokers can suffer.
Third, the FDA authorized Vuse may let Vuse’s market share soared. Nielsen data for the week ending 9/11 showed Juul Labs’ U.S. e-cigarette market share at 40.9%, followed by Anglo American’s Vuse at 32.3%. In third place was NJOY Holdings Inc.’s NJOY brand with 3.5%.
The data shows that Vuse is already closing in on Juul’s market share, and if Juul’s license is delayed and Vuse makes further strides, Juul’s position is in jeopardy.
The U.S. e-cigarette policy will be a great reference for the global regulation of e-cigarettes, which can be said to be the vane of the e-cigarette market.
FDA’s first e-cigarette authorization, the entire e-cigarette industry is a good thing, meaning that e-cigarettes have a legal identity, Strict regulation is not a bad thing, a broad brush is the most terrible.


